Name: Lenika
Partner(s) Names: Kenia
Date (the experiment was performed, NOT the due date of the report): 9/3/15
Purpose:
Our goal for this experiment is to study the flame color reaction to different types of ionic compounds.
Procedure:
With a dry watch glass, methanol will be dripped onto to different ionic compounds then lit on fire to create a small flame. The flame color will then depend on the different ionic compounds that are going t be tested in this experiment.
Pre-Lab Questions:
What is making color in a flame test?
Metal ions reaction excites the electron and the energy in a photon determines the flames color.
What color of light is the lowest in energy?
Red is the lowest in energy.
What color of light is the highest in energy?
Violet has the highest amount of energy.
What color of light is the highest frequency?
Violet has the highest frequency.
What color of light is the lowest frequency?
Red has the lowest frequency.
In chemistry, what is a salt?
Salt is ionic compounds in chemistry.
How are electrons “excited”?
They're showing excitement by jumping and creating wavelengths.
What is a “ground state”?
When all of the electrons are at the lowest possible energy level.
In your own words, write a short explanation of how an electron absorbs energy and re-emits it as light and why different elements have different spectra.
Electrons absorb energy and become excited and re-emit it by shooting a photon out to lose their energy.
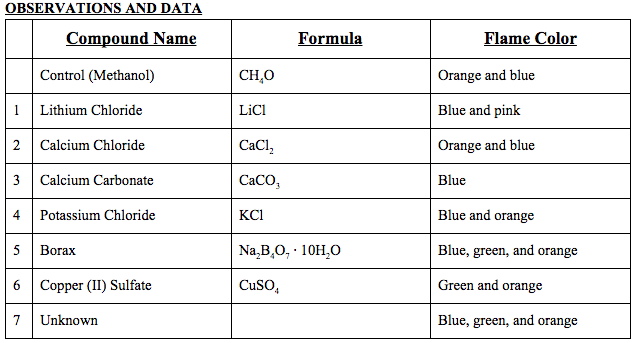
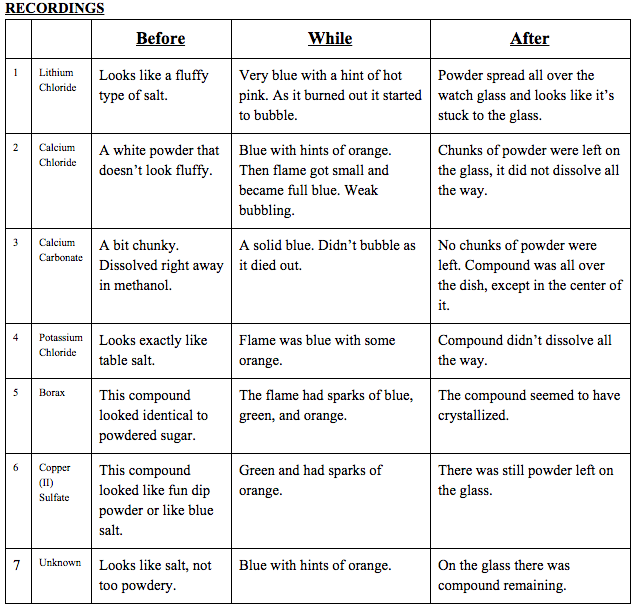
Data Table(s):
(The unknown was borax.)
Partner(s) Names: Kenia
Date (the experiment was performed, NOT the due date of the report): 9/3/15
Purpose:
Our goal for this experiment is to study the flame color reaction to different types of ionic compounds.
Procedure:
With a dry watch glass, methanol will be dripped onto to different ionic compounds then lit on fire to create a small flame. The flame color will then depend on the different ionic compounds that are going t be tested in this experiment.
Pre-Lab Questions:
What is making color in a flame test?
Metal ions reaction excites the electron and the energy in a photon determines the flames color.
What color of light is the lowest in energy?
Red is the lowest in energy.
What color of light is the highest in energy?
Violet has the highest amount of energy.
What color of light is the highest frequency?
Violet has the highest frequency.
What color of light is the lowest frequency?
Red has the lowest frequency.
In chemistry, what is a salt?
Salt is ionic compounds in chemistry.
How are electrons “excited”?
They're showing excitement by jumping and creating wavelengths.
What is a “ground state”?
When all of the electrons are at the lowest possible energy level.
In your own words, write a short explanation of how an electron absorbs energy and re-emits it as light and why different elements have different spectra.
Electrons absorb energy and become excited and re-emit it by shooting a photon out to lose their energy.
Data Table(s):
(The unknown was borax.)
Observations:
Conclusion:
Overall, the purpose of this experiment was completed.
Discussion of Theory:
The flame test is a procedure used to test quantitatively for the presence of certain metals in chemical compounds. This means that it is a procedure that includes lighting ionic compounds on fire, to see what color they turn once lit up. This is how it works, the compound is heating up, which causes the electrons within the compound to absorb energy. They then jump into an excited energy state, or a quantum jump. They don’t stay excited for long, and after a while they fall back into their ground state; like a sugar crash. When the electrons lose their energy they will emit a photon of light. The photons emitted will match the quantum energy difference between the excited state and ground state, making the colors appear.
Error Analysis:
My partner and I didn't not make a mistake and we did not run into any issues. I guess if I have to be nit-picky, I would say that I once placed my lab notebook on the desk while the group across from us was running their experiment.
Post Lab Questions:
Why is it important to test the flame color of the methanol without any compounds dissolved in it?
It is important because you have to know the color of the methanol because when you test the compounds, you don't want to confuse the color of the methanol with the flame of the compound.
List the colors observed in this lab from highest energy to lowest energy.
Violet, Indigo, Blue, Green, Yellow, Orange, Red.
List the colors observed in this lab from highest frequency to lowest frequency.
Violet, Indigo, Blue, Green, Yellow, Orange, Red.
List the colors observed in this lab from shortest wavelength to longest wavelength.
Violet, Indigo, Blue, Green, Yellow, Orange, Red.
What is the relationship between energy, frequency, and wavelength?
They all use the same color spectrum (Red, orange, yellow, green, blue, indigo, and violet), which is then reversed to form the answers for the color spectrum of the flames.
Based on the results of your experiments, what metal was found in your unknowns? Explain.
I think this because I saw that the flame was green, yellow, red , and blue. Yellow and blue could account for the color of the flame. If we eliminate those colors, we have green and red. The green could be accounted for by the chemical Copper Nitrate (Cu(NO3)2 and the red can be accounted for by the Lithium Chloride (LiCl).
Do you think we can use the flame test to determine the identity of unknowns in a mixture? Why or why not?
I think we can use the flame test to find out the unknowns. I think this because I think we can figure out the unknown by the color of the flame. Then, we can find out by subtracting the normal colors that would normally be in a flame such as yellow and blue. Then, we can find out the other colors and guess the unknown compound.
Why do different chemicals emit different colors of light?
I think that different chemicals emit different colors of light because of their frequency, wavelength and energy.
Most salts contain a metal and a non-metal. Look at the compounds we tested and explain how we can be sure that it is the metal atoms that are responsible for the colors that you see.
The metal atoms contain the reactive particles, therefore making the colors in the flame.
Colorful light emissions are applicable to everyday life. Where else have you observed colorful light emissions? Are these light emission applications related? Explain.
I have observed colorful light emissions in fireworks shows and videos of Auroras. The fireworks show is because they use chemicals to produce the different colors and an Aurora is because of the metals and gases in the atmosphere colliding with solar particles.
Can you think of a way in which to use the flame test? Please describe below.
I think the flame test may be used to explore many areas of exploration and research such as solar particles.
Overall, the purpose of this experiment was completed.
Discussion of Theory:
The flame test is a procedure used to test quantitatively for the presence of certain metals in chemical compounds. This means that it is a procedure that includes lighting ionic compounds on fire, to see what color they turn once lit up. This is how it works, the compound is heating up, which causes the electrons within the compound to absorb energy. They then jump into an excited energy state, or a quantum jump. They don’t stay excited for long, and after a while they fall back into their ground state; like a sugar crash. When the electrons lose their energy they will emit a photon of light. The photons emitted will match the quantum energy difference between the excited state and ground state, making the colors appear.
Error Analysis:
My partner and I didn't not make a mistake and we did not run into any issues. I guess if I have to be nit-picky, I would say that I once placed my lab notebook on the desk while the group across from us was running their experiment.
Post Lab Questions:
Why is it important to test the flame color of the methanol without any compounds dissolved in it?
It is important because you have to know the color of the methanol because when you test the compounds, you don't want to confuse the color of the methanol with the flame of the compound.
List the colors observed in this lab from highest energy to lowest energy.
Violet, Indigo, Blue, Green, Yellow, Orange, Red.
List the colors observed in this lab from highest frequency to lowest frequency.
Violet, Indigo, Blue, Green, Yellow, Orange, Red.
List the colors observed in this lab from shortest wavelength to longest wavelength.
Violet, Indigo, Blue, Green, Yellow, Orange, Red.
What is the relationship between energy, frequency, and wavelength?
They all use the same color spectrum (Red, orange, yellow, green, blue, indigo, and violet), which is then reversed to form the answers for the color spectrum of the flames.
Based on the results of your experiments, what metal was found in your unknowns? Explain.
I think this because I saw that the flame was green, yellow, red , and blue. Yellow and blue could account for the color of the flame. If we eliminate those colors, we have green and red. The green could be accounted for by the chemical Copper Nitrate (Cu(NO3)2 and the red can be accounted for by the Lithium Chloride (LiCl).
Do you think we can use the flame test to determine the identity of unknowns in a mixture? Why or why not?
I think we can use the flame test to find out the unknowns. I think this because I think we can figure out the unknown by the color of the flame. Then, we can find out by subtracting the normal colors that would normally be in a flame such as yellow and blue. Then, we can find out the other colors and guess the unknown compound.
Why do different chemicals emit different colors of light?
I think that different chemicals emit different colors of light because of their frequency, wavelength and energy.
Most salts contain a metal and a non-metal. Look at the compounds we tested and explain how we can be sure that it is the metal atoms that are responsible for the colors that you see.
The metal atoms contain the reactive particles, therefore making the colors in the flame.
Colorful light emissions are applicable to everyday life. Where else have you observed colorful light emissions? Are these light emission applications related? Explain.
I have observed colorful light emissions in fireworks shows and videos of Auroras. The fireworks show is because they use chemicals to produce the different colors and an Aurora is because of the metals and gases in the atmosphere colliding with solar particles.
Can you think of a way in which to use the flame test? Please describe below.
I think the flame test may be used to explore many areas of exploration and research such as solar particles.






 RSS Feed
RSS Feed
